Direct COVID-19 RT-qPCR Kit – No RNA Extraction
DIRECT RT-PCR Amplification, Eliminates The Difficult RNA Extraction Step
Sample To Results In Only 60 minutes
This kit is used for the detection of the coronavirus (COVID- 19 RT PCR Kit). Samples can be obtained from throat swab or sputum.
Catalog Number: MXS3
Size: 96 tests/rxns
Also Known As: COVID-19, SARS-CoV-2, Direct RT-qPCR, ORF1ab, N
Direct RT-PCR Amplification
No RNA Extraction Required! We use MaxBuffer Tube that skips the difficult RNA Extraction Process.
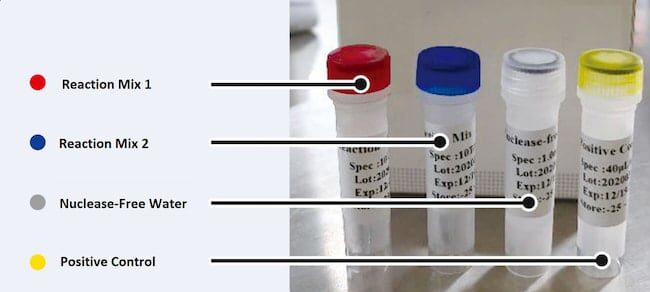
This kit includes all componets (Primers, Probes, Positive, Negative Control etc.). MaxBuffer Tube should be bought separately.
Price Comparison of COVID-19 RT-qPCR Kits
| COMPANY | RXNS | PRECIO en $us. |
| Biovendor | 100 | $us. 975 |
| Geneon | 100 | $us. 761.98
(699 Euros) |
| Eurofinsgenomics | 96 | $us. 1396 |
| MaxPrecision Lab | 96 | $us. 700* |
We offer huge discounts for orders >50,000reactions.
The price of this kit doesn’t include MaxBuffer Tube

Our COVID-19 RT-qPCR Kit Is Fully Compatible With:
-
- ABI 7500
- Bio-Rad CFX96, CFX384
- Bio-Rad Touch / iQ5
- Cepheid Smartcycler/Smartcycler II
- 7 QuanStudio’s 6, 7, 12k
- Roche: LightCycler®480/1536/Nano
- Agilent: Mx3005p, Mx3000P
- Qiagen: RotorGene6000,
- QEppendorf Mastercycler
Componests MaxSars COVID-19 RT-qPCR Kit:
| Part | Volume (μl/Vial) 96T Kit |
| Reaction Mix | 1600 μL |
| RT Enzyme Mix | 46 μl |
| Positive control | 40 μl |
| Nuclease-Free Water | 1.0 ml |

Componests MaxSars COVID-19 RT-qPCR Kit:
| Specifc Genes | Fluorecent Dyes | |
| Gene 1 | ORF1ab (RdRp) | FAM |
| Gene 2 | N Gene | VIC |
| Internal Control | RNase P | CY5 |
Reaction Mix are basic components of this detection The reaction mixes in this kit contain specific primers and probes for N gene and ORF1ab of COVID-19, and RNase P gene as sample internal control (IC), as well as all components needed for efficient RT-PCR reactions.
The positive control is the mix of single-stranded RNA of specific regions of novel coronavirus (COVID-19), required to be dispensed into 200 μL PCR tubes with 5μL per reaction according to the required number of reactions. Please avoid repeated frozen-thawed cycles to avoid the degradation of RNA templates.
Components from different batch number COVID-19 RT-PCR kits are not recommended to be used interchangeably.

Materials and Devices Required but Not Provided:
- Biological Cabinet
- Appropriate Viral RNA Extraction Kit or MaxBuffer Tube
- Desktop centrifuge (suitable for 96-well plate or 8-strip tube).
- Centrifuge with a rotor for 2ml reaction tubes or plate.
- Vortex
- Adjustable pipetts (with maximum capacity of 2μL, 10μL, 50μL, 100μL, and 200μL, respectively)
- Disposable pipette tips with filters
- Disposable powder-free
- RNase free 1.5mL centrifuge
- PCR reaction tube / PCR reaction
NOTE: Please ensure that the PCR instrument and materials have been installed, calibrated, checked, and maintained according to the manufacturer’s instructions and recommendations.
Procedure:
- Release or Extract the RNA sample with an RNA Extraction kit or with MaxBuffer Tube.
- Formulation of RT-PCR One-step
-
- Determine the amounts of samples to be tested first; RNase-free reaction tubes shall be provided with each
- Thaw Reaction Mix, positive control, and RNA samples on the ice, and shake with inching on the shaker, followed by short spin on the
- Reaction mixes contain RT-PCR primers, probes and reaction reagents (except enzymes). Primers and probes for Internal control (IC) gene have been included in the reaction Set up following ingredients in order:
-
Table, Detection of each assay (example with reactions for ORF1ab, N gene and IC gene).Add the components as the order below:
| Reagents | Individual test (μL) |
| Reaction Mix (ORF1ab/N gene/IC) | 14.6 |
| RT Enzyme Mix | 0.4 |
| RNA sample | 5 |
| Total volume | 20 |
- Add the 14.6 μL Reaction Mix and 0.4 μL RT Enzyme Mix together. Calculate the volume (μL) needed: number of tests multiply Reaction Mix or RT Enzyme Mix amount; prepare 10% more.
- Aliquot 15 μL of final Reaction Mix into each sample
- The total RT-PCR reaction volume is 20 μL:
-
- RNA sample: Add 5μL of RNA sample to the corresponding reaction well in Reaction Mix.
- Positive control: Add 5μL of positive control to the reaction well in Reaction Mix as positive control well.
- Negative control: Add 5μL of Nuclease-free water to the reaction well in Reaction Mix as negative control well.
- Seal the tube cap and shake with inching on the shaker several times, followed by short spin on the centrifuge.
- RT-PCR protocol as below:
| Temp (℃) | Time | Cycles |
| 50 | 5 min | 1 |
| 95 | 20 sec | 1 |
| 95 | 5 sec |
45 |
| 60 | 30 sec |
Probe label:
ORF1ab: FAM-BHQ1; N Gene: VIC-BHQ1; IC (RNase P) tube: CY5- BHQ3.
- Result Interpretation
-
- Quality Check for the Test Results
The following requirements on value Ct of positive control well and negative control well on the reaction plate within the same reaction plate/batch:
| Quality control requirement | |
| Positive Control reaction well | Ct≤37 |
| Negative Control reaction well | Ct>37 |
- The experiment is invalidated, and repeat is required if the positive control and/or negative control does not meet the criteria set above.
- The analysis of the Ct value of the ORF1ab, N, and IC wells in each swab or sputum specimen as follows:
| ORF1ab | N gene | IC | Interpretation |
| Ct≤37 | Ct≤37 | Ct≤40 | Positive |
| Ct>40 or Undet | Ct>40 or Undet | Ct≤40 | Negative |
|
40>Ct>37 (Amplifiable) |
40>Ct>37 (Amplifiable) |
Ct≤40 |
Weak positive; Retest
to confirm |
| Ct>40 or Undet | Ct≤37 | Ct≤40 | Coronaviral infection |
|
Ct>37 or Undet |
Ct>37 or Undet |
Ct>40 or undet |
Resampling for test or other confirmation |
| Ct≤37 | Ct>37 or Undet |
Performance Evaluation:
- Limit of Detection (LOD): 400 copies/mL.
- Interference reaction : The five potential reference(Dexamethasone, Azithromycin, Tobramycln, Levofloxacin, Ceftriaxone) will not interfere with the detection results of the kit.
- Cross-reavity: No cross reaction with 16 viruses(Human BK polyomavirus, Human adenovirus C serotype 5, Human adenovirus A/B1/C/D/E, Human herpesvirus 1/2/3/4/5/7, Human parvovirus B19, Human JC polyomavirus, Simian vacuolating virus 40) and human genome DNA
- Internal precision: repeatability : cv% < 10%, between-run precision : cv% < 10%, between- day precision : cv% < 10%, total precision: cv% < 10%..
- External precision: repeatability : cv% < 10%, between-run precision : cv% < 10%, total precision: cv% < 10%.
Performance Limitation:
Test results only serve as clinical reference and comprehensive judgments based on clinical symptoms and other laboratory tests method should be considered by clinicians.
- Negative results cannot completely rule out the existence of novel Improper sample collection, improper transportation, improper processing and insufficient initiation VL(viral load) may influences the experimental results.
- Other unverified interferences or PCR inhibitors may cause false negative results.
Warnings and Precautions:
- Carefully read this instruction before starting the procedure.
- Components from different batch number kit cannot be used interchangeably.
- Once each component within the kit is thawed, it is suggested to use them up within one operation based on examinaton demand, and the component remained should be restored at -20℃. Repeated freezing and thawing (more than three times) of reagents should be avoided.
- Viral RNA and RT-PCR premix are sensitive to temperature. Once Sample RNA and RT-PCR pre-mix are taken out of -20±5℃ freezer, prepare the MasterMix on ice or in the cooling block.
- Avoid microbial and nuclease (DNase/RNase) contamination of the specimen and the components of the kit.
- Always use DNase/RNase-free disposable aerosol-blocking pipette tips.
- Use of this product is limited to personnel specifically instructed and trained in the Wear protective disposable powder-free gloves, laboratory coat and eye protection when handling specimens and kit reagents.
- Use separated and segregated working areas for (i) reaction set-up, (ii) specimen preparation and (iii) amplification/detection activitie Workflow in the laboratory should proceed in unidirectional manner. Wear separate coats and gloves in each area.
- All biological samples and materials with the contact with the product should be treated as infectious biohazard, and related local regulations shall be followed for the Prevent the exposure to skin and mucosa.
- The detection test with this kit should be conducted by medical staff and technician with professional technical training.
- Store positive and/or potencially positive material separated from all other components of the kit.
- Do not open the reaction tubes/plate post amplification to avoid contamination with amplicons.
- Additional controls may be tested according to guidelines or requirements of local, state and/or federal regulations or accrediting organizatio
- Discard sample and assay waste according to your local safety regulations.
- Do not eat, drink, or smoke in the laboratory working area.
- Do not use components of the kit after expiration date.

Our Production Team Has Been Working Very Hard To Fulfill The High Demand For Covid-19 RT-PCR Kits.
Therefore The Number Of Samples Is Limited.
In Order To Confirm The Number Of Samples Available. Please Send Us An E-Mail To: info@maxprecisionlab.com
MaxSars COVID-19 RT-qPCR Kit Follows Recommendations From The WHO And US CDC.
- High Specificity For Target Markers ORF1ab , N.
- Fast Results, In Less Than 1 Hour.
- Possitive, Negative Control, Primers, Probes Included.
MaxPrecision Lab Warranties that all products will be FREE from defects in materials, workmanship and will perform in conformance with our protocols*.